Advantages of Titanium Rods as Orthopedic Implants
Medical device manufacturers and buyers seek safe, durable, and cost-effective materials for joint usage. Titanium rod surgical implants are biocompatible and mechanically superior to other options. These precision-engineered components tackle spine fixation, fracture stabilisation, and joint repair issues where material failure might slow patient recovery. Titanium's natural oxide coating prevents tissue damage and has a greater strength-to-weight ratio than most medical steels. Pure and alloyed titanium bars that satisfy ASTM F136 and ISO 5832-3 standards are being used in the worldwide orthopaedic implant market. These guidelines maintain the quality of life-changing therapy.

Understanding Titanium Rods in Orthopaedic Implants
Orthopaedic implants employ titanium rods. To acquire hip implants, you must first grasp the differences between medical-grade and industrial titanium. Titanium's atomic structure forms a passive layer that repairs immediately when fractured. It resists biological corrosion totally. Since implants remain exposed to saline and mechanical stresses for decades, this quality is crucial.

Chemical Composition and Mechanical Properties
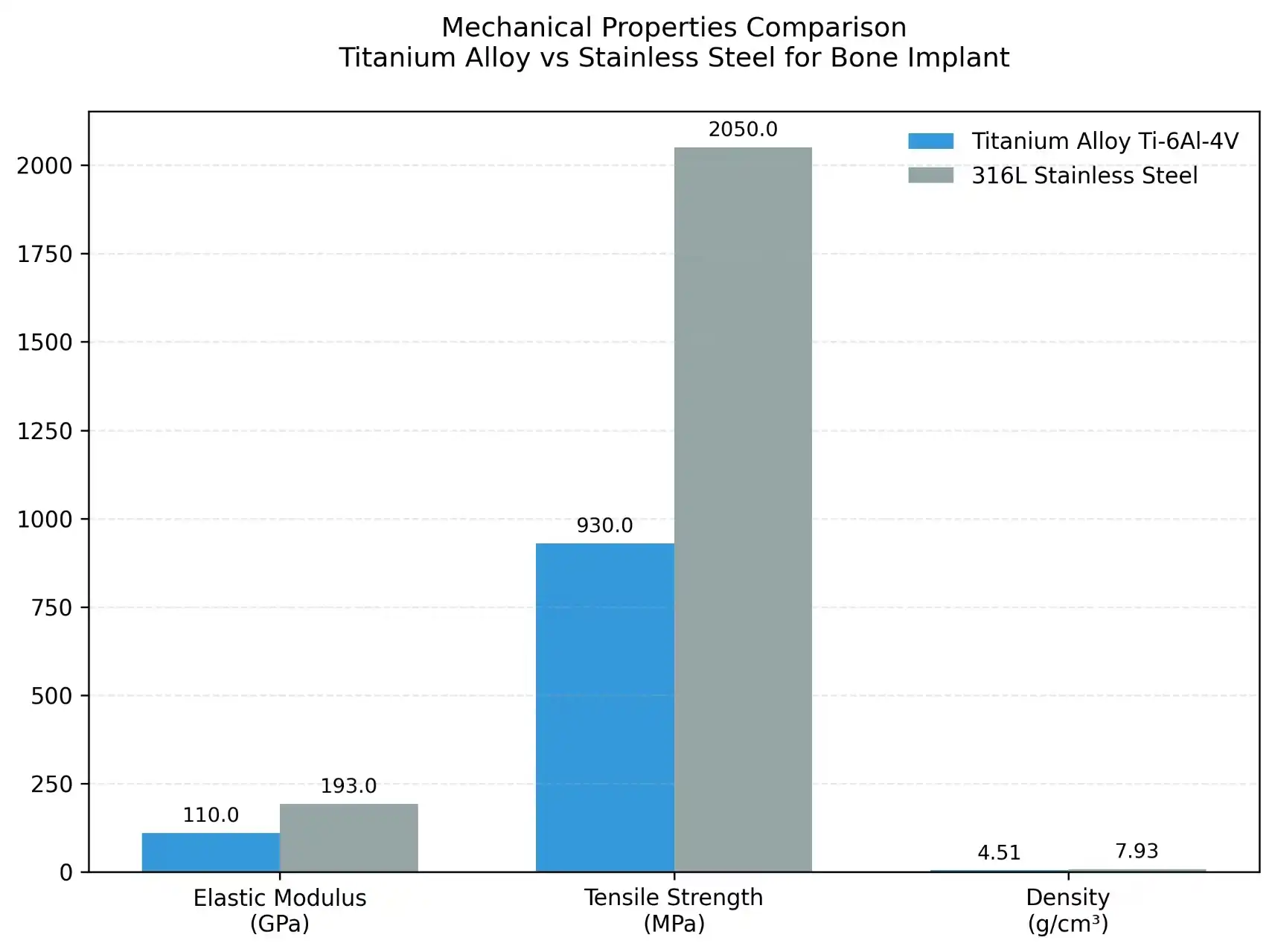
Medical-grade titanium comes in alloys and commercially pure grades (1–4). Grade 2 titanium, 99.2% pure titanium with little oxygen and iron, is robust and rust-proof. Its 345 MPa tensile strength is sufficient for many medical applications that need a light load. Implants often employ titanium grade 5 (Ti-6Al-4V). The 6% aluminium and 4% vanadium alloy has over 895 MPa tensile strength. This alloy is for high-stress places like hip stems and spine plates, where failure would be disastrous.
Titanium is more like bone than stainless steel because its elastic stiffness is 110 GPa compared to 200 GPa. This fit reduces stress shielding, which happens when stiff implants prevent bone remodelling, causing bone loss and implant loosening. Purchasers should review mechanical testing data to validate these attributes when choosing suppliers, as processing might alter product performance.
Manufacturing Processes for Medical-Grade Quality
From raw titanium paper to authorised medicinal bars, the procedure is lengthy and delicate. Air pollution is removed by vacuum arc remelting, creating uniform ingots without oxygen patches that may break. Forging at regulated temperatures smooths the grain structure, making it wear-resistant. Implants are loaded and unloaded millions of times over a patient's lifetime, making this crucial.
Hot rolling or rotational forging reduces breadth without changing the metal structure. Centerless grinding achieves diameter variances of ±0.05mm, ideal for surgical instruments and implant pieces. Distinct surface treatments serve distinct purposes. Sanding creates decorative mirror finishes, while pickling removes acid scale for coatings. Annealing heat decreases metal tensions and optimises strength and flexibility.

Baoji Jucheng Titanium Industry makes 6mm to 450mm bars using vacuum melting and precision forging technologies refined over 20 years. Special surgical systems may use lengths up to 12000mm, although common lengths are 6000mm. These sizes accommodate anything from bone pins to spine stabilisation rods. Following ASTM B348, ASME SB348, and AMS 4928 requirements throughout manufacturing ensures that every batch satisfies global medical device producers' demands.
Core Advantages of Titanium Rods in Orthopaedic Applications
Choosing implant materials requires balancing biological safety, mechanical performance, and long-term reliability. Modern orthopaedic surgery favours titanium bars because they suit all of these criteria better than other metals.
Superior Biocompatibility and Osseointegration
Chemically neutral titanium has a self-growing oxide layer, making it biocompatible. Titanium has a less immunological response than nickel or cobalt, reducing rejection and inflammation. Human studies demonstrate that titanium implants may integrate with tissue at above 95% in adequately prepared surgical sites.
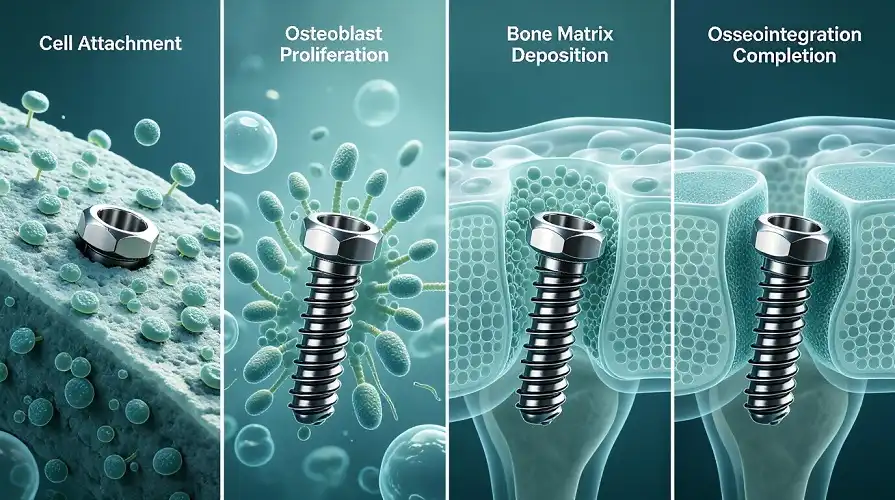
Titanium helps osteoblasts stay and proliferate, allowing bone tissue to immediately adhere to the implant surface. Titanium rods help bone develop over spine fusion sites, stabilising the structure. Hip replacement stems constructed of titanium metal bars develop bone that secures the implant without cement. This biological link prevents implant failure and discomfort from micromotions. This implies that many patients' implants will survive over 20 years and perform perfectly. The CP titanium rod is ideal for such applications because of its unmatched biocompatibility.
Biocompatibility testing, including cytotoxicity screens and sensitisation studies, must be checked when medical device producers receive raw materials. Extra-low interstitial grade 23 (Ti-6Al-4V ELI) has less oxygen and iron. It is excellent for load-bearing usage since permanent implants are biocompatible.
Mechanical Strength and Corrosion Resistance
Hip implants undergo several mechanical stressors at various applications. The patient's weight must twist intramedullary pins that keep femoral fractures together, and spine rods must withstand torsional stresses as the patient twists and bends. Titanium alloy bars are 45% lighter than stainless steel yet have the same tensile strength. They reduce tissue tension surrounding the implant and enable smaller forms.
Titanium products last longer, making them stand out. Grade 5 bars may resist over 10 million loading cycles at human stress levels without breaking if properly handled. This lifespan is crucial for active persons who stress their implants all day. The final performance feature is corrosion prevention. In chloride-rich bodily fluids that pit or stress-corrode stainless steel, titanium remains stable. This defence stabilises dimensions and prevents organ metal ion buildup.

Titanium outperforms other materials in comparison. While cheaper, stainless steel is more prone to corrosion and may cause nickel allergies. Aluminium is too weak for load-bearing. Carbon fibre composites have a low modulus but are difficult to produce, and their biological effects are unknown. Titanium is the ideal option for risk-averse purchasing techniques since it is well-regulated and proven.
Case Examples in Spinal and Hip Implants
Titanium bars are challenging to utilise in spinal stabilisation devices. Several spinal segments are connected by 5mm–6mm rods for posterior fusion. Over the following 6–12 months, these rods must withstand compressive, stretching, and twisting pressures as the bone fuses. Grade 5 titanium rods are robust but not inflexible, which might stress adjoining spine segments and induce adjacent segment illness.
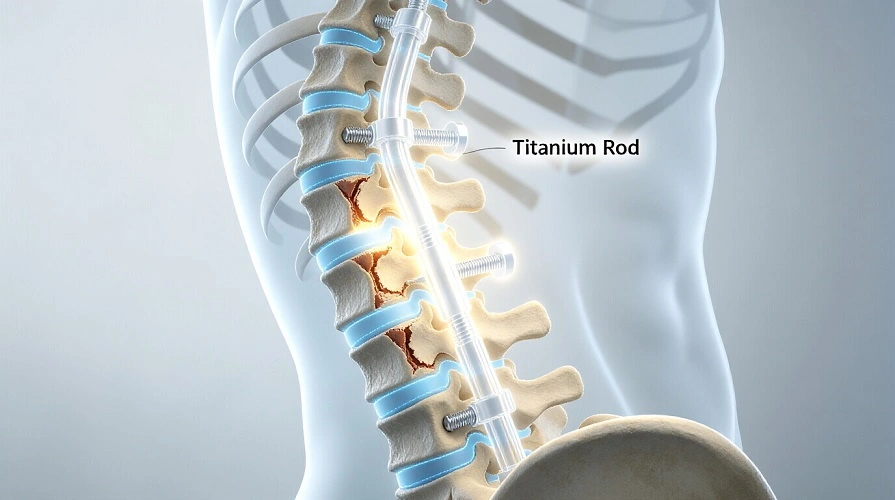
Titanium stems from thick bars are used in more hip replacement systems. Modular designs allow surgeons to adjust length and angle to accommodate a variety of patients. To promote bone growth, plasma spray or bead sintering coatings make titanium surfaces porous. This makes biological mending better than cement. Acetabular cups function similarly. Thin sides assist bone fusion, and titanium shells sustain the structure.

Selecting the Right Titanium Rod for Orthopaedic Implants
Buying things has an impact on how well they work, how well they follow the rules, and finally, how well patients do. To do strategic sourcing, you need to know about grade requirements, the skills of the seller, and the total cost of ownership.
Titanium Grades for Medical Applications
varied commercially pure titanium has varied mechanical properties. The softest and most mouldable grade 1 is excellent for non-weight-bearing items like medical instrument handles. Grade 2 is strong and workable, making it ideal for trauma stabilisation plates and pins. Grade 4, with more oxygen, is stronger but less flexible; it can't be utilised for bendy items.
Grade 5 (Ti-6Al-4V) is the most preferred metal because of its strength-to-weight ratio and medical history. Acquisition teams should be aware that aluminium and vanadium alloys raise concerns about their long-term consequences on living organisms, thus some manufacturers are exploring alternate metals. Grade 23 (Ti-6Al-4V ELI) has stricter chemical restrictions that eliminate interstitial elements that make the material less flexible and incompatible with living organisms. Long-lasting implants justify the increased price of this grade.
Palladium-containing Grade 7 and Grade 12 prevent crevice corrosion in severe conditions. In orthopaedics, these grades are seldom utilised; they may be appropriate for disinfectant-exposed exterior bracing devices. Grade 9 (Ti-3Al-2.5V) tubing for minimally invasive surgical tools is intermediately strong due to its lower alloy concentration.
Supplier Certification and Quality Standards
Obtaining medical supplies requires regulatory compliance. Suppliers of surgical implants must meet ASTM F136, which regulates chemical composition, mechanical property criteria, and testing. ISO 5832-3 simplifies global supply chains with international equivalency. ASME SB348 sets titanium bar specifications, while AMS 6931 covers aerospace-grade material that exceeds medical demands.
Medical equipment manufacturers should maintain ISO 13485 accreditation, which verifies their quality control processes. FDA registration doesn't guarantee items, but it shows a provider understands the requirements. Material tracking enables you to respond fast if quality issues develop by following each bar from the melt batch to shipping.
Baoji Jucheng Titanium Industry has several licenses and collaborates with Tsinghua University and the Northwest Nonferrous Metals Research Institute to increase quality. The company's testing capabilities include spectroscopy for chemical analysis, tensile and impact tests for mechanical qualities, and metallography for microstructural integrity. For medical device businesses that preserve design control files, this unified solution ensures batch-to-batch consistency.
Cost-Benefit Analysis for Procurement
Initial material costs are just a portion of ownership costs. Titanium rod bars cost three to five times more per kilogram than stainless steel bars, depending on quality and market. When considering titanium's improved machinability, this price drops. Titanium reduces cycle times and tool wear. Lighter packages save money on shipping due to decreased density. It allows smaller implant designs, reducing operation harm.
Low review ratings indicate long-term worth. Implant failures require costly second procedures, put companies at risk of lawsuits, and damage brand image. Titanium's long life and ability to cooperate with living things reduce these dangers, benefiting patients and companies. Procurement strategies should include failure cost models. This implies paying for issues that might have been prevented with high-quality materials.
Bulk purchases with well-known vendors may provide economies of scale. Working with stockists may help smaller producers reach minimum order volumes. Jucheng Titanium has 3,000 tonnes of stock in various grades and sizes to accomplish works fast without requiring clients to transfer too much.
Procurement and Supply Chain Considerations for Titanium Rods
Effective buying includes more than just knowing what materials to buy. It also includes managing relationships with suppliers, coordinating supplies, and lowering risks.
Evaluating Supplier Reliability and Capabilities
First, evaluate a supplier's manufacturing capabilities. Can the supplier always meet deadlines? The 120,000-square-meter Baoji Jucheng Titanium factory offers full processing capabilities, from vacuum melting to surface treatment. The firm doesn't need outside processors, which might delay or lower the quality. Over 1,500 tonnes of particular titanium items are produced annually, indicating the firm can support additional medical device activities.
Technical support distinguishes strategic sellers from transactional suppliers. People creating medical devices typically require aid with material regulations, alloys, and material qualities. Suppliers with research and development teams, like Jucheng's network of universities and research institutions, may provide this advice. The firm owns 4 innovation patents and 41 utility model patents, demonstrating its commitment to improving processes and goods for consumers.
Long-term quality consistency is as crucial as initial quality. Check your suppliers' quality records for rejections, non-conformance, and corrective action responses. Suppliers with decades of experience have more reliable processes than newer ones. Jucheng's 20 years of titanium handling and designation as a National High-Tech Enterprise and "little giant" inspire confidence in its long-term performance.
Global Sourcing and Lead Time Management
Most titanium is found in the aerospace and defence industries. US, Russian, and Chinese manufacturing dominate the globe. Secondary processing and bar manufacturing are possible in India, Europe, and Southeast Asia. When choosing a supplier, evaluate lower prices versus logistical and government constraints.
Order quantity, quality, and size determine lead times. In stock grades like Grade 2 and Grade 5 may ship within weeks, while bespoke sizes or specialised grades like Grade 23 ELI can take 8 to 16 weeks to collect the raw ingredients, process them, and ensure quality. Rush orders cost extra and may violate patient safety standards.
Supply chain issues may be resolved by knowing local merchants. Wholesalers charge more and may not know how to address application-specific issues. Working directly with manufacturers like Jucheng, which has strengthened worldwide export channels via ties with overseas customers like Jiangxi Copper Group, saves money and provides technical knowledge.
Customisation and OEM Specifications
Differentiating medical devices often needs custom material solutions. Precision fits are possible in modular implant systems thanks to custom circle limits that are tighter than standard requirements. For optimal practical performance, surfaces should be either polished for visual purposes or roughened for osseointegration purposes. For some uses, different types of heat treatment are needed to change the hardness or residual stress patterns.
By taking care of these changes, suppliers that offer OEM/ODM services speed up the creation process. Jucheng Titanium can make more than just bars. They can also make finished implant parts using CNC machining centres, which can turn raw bars into parts that are very close to a net shape. The number of supply chain handoffs is cut down by this vertical integration. Each handoff adds a chance for quality problems and takes longer than planned. Purchasing teams can combine their ties with vendors, which makes quality checks and regulatory paperwork easier.

Conclusion
Titanium rods excel in hip implant uses because they are biocompatible, strong, and resistant to corrosion, making them better than other materials. When buying these important parts for medical devices, people who make them and people who do the buying should know about grade standards, source capabilities, and total cost. Having strategic relationships with well-known providers that offer full certifications, technical support, and reliable supply lines lowers risks and encourages new ideas. Titanium's place in orthopaedic medicine will continue to grow as processing technologies improve and concerns about sustainability grow. Purchasing teams that build knowledgeable, flexible buying strategies now will be rewarded in the future.
FAQ
1. What titanium grades are most suitable for permanent orthopaedic implants?
Grade 23 (Ti-6Al-4V ELI) is ideal for long-term load-bearing implants like hip stems and spinal plates because of its high biocompatibility and toughness. Grade 2 pure titanium is better for non-load-bearing implants such as plates and screws, where corrosion resistance is more important than strength. The best choice depends on load requirements, implantation time, and regulatory needs.
2. How do processing methods affect medical-grade titanium bar quality?
Vacuum melting reduces contaminants, while controlled forging improves grain structure and mechanical performance. Annealing balances strength and flexibility, which is critical for surgical use. Precision finishing methods like centerless grinding ensure tight tolerances for implant manufacturing. Suppliers handling all processes in-house usually provide more consistent quality and better traceability.
3. What certifications should I verify when selecting a titanium rod supplier?
Check for ASTM F136 compliance for surgical implant materials, ISO 13485 certification for medical quality management, and ISO 5832-3 standards for implant-grade titanium. Request material test reports for each batch, including tensile strength and chemical analysis, and ensure the supplier offers full traceability from melting to shipment.
Partner with Jucheng Titanium for Your Medical-Grade Titanium Rod Needs

It has been making fine titanium bars for medical and surgical uses for more than 20 years, according to Baoji Jucheng Titanium Industry Co., Ltd. To meet your exact needs, our factory makes materials from Grade 1 to Grade 23 ELI, with widths from 6mm to 450mm and lengths of up to 12000mm. We have ASTM F136, ISO 5832-3, and ASME SB348 certifications, along with a lot of paperwork that shows how they were earned, and every titanium rod supplier relationship we make benefits from them. We keep 3,000 tons of goods on hand all year, so we can quickly turn around even large orders. Our R&D partnerships with top research schools give us access to the newest processing technologies. Email our expert team at s4@juchengti.com to talk about how our OEM/ODM services and quality assurance tools can help your medical device supply chain.
References
1. Long, M. and Rack, H.J., "Titanium Alloys in Total Joint Replacement—A Materials Science Perspective," Biomaterials, Vol. 19, 1998, pp. 1621-1639.
2. Niinomi, M., "Mechanical Properties of Biomedical Titanium Alloys," Materials Science and Engineering A, Vol. 243, 1998, pp. 231-236.
3. Steinemann, S.G., "Metal Implants and Surface Reactions," Injury, International Journal of the Care of the Injured, Vol. 27, Supplement 3, 1996, pp. C16-C22.
4. Rack, H.J. and Qazi, J.I., "Titanium Alloys for Biomedical Applications," Materials Science and Engineering C, Vol. 26, 2006, pp. 1269-1277.
5. Brunette, D.M., et al., "Titanium in Medicine: Material Science, Surface Science, Engineering, Biological Responses and Medical Applications," Springer-Verlag Berlin Heidelberg, 2001.
6. Geetha, M., et al., "Ti-Based Biomaterials, the Ultimate Choice for Orthopaedic Implants—A Review," Progress in Materials Science, Vol. 54, 2009, pp. 397-425.

