Are titanium plates used in open-heart bypass surgery
However, titanium plate parts are not used in bypass grafts themselves. They are only used in certain heart surgeries. Most of the time, these bendable plates are used to close the sternum and give extra support during open-heart treatments. Titanium metals made for medical use, especially Ti-6Al-4V, don't rust and work well with tissues. They are very useful for supporting important heart treatments because they keep patients safe and help them heal quickly.
Understanding Titanium Plates in Medical Applications
Titanium goods are famous in medicine because they are strong, don't react with living things, and don't rust. Surgical metals like stainless steel and cobalt-chromium alloys are not the same as medical-grade titanium because they work better.

Biocompatibility and Safety Standards
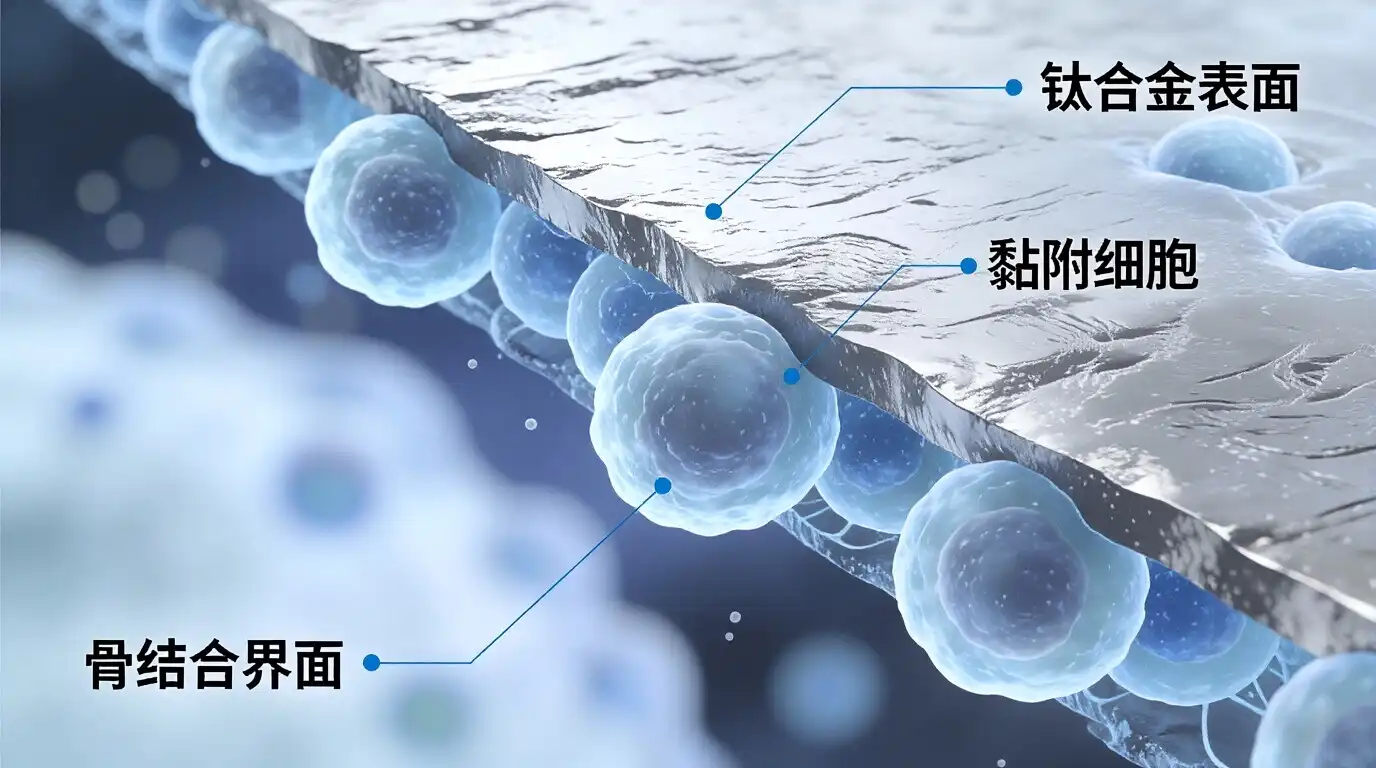
Medical titanium plate is put through a lot of tests to meet international standards such as ASTM F67 and ISO 5832-2. If you follow these rules, your titanium implants will stay neutral in your body. This will stop any bad responses in the tissue that could slow down the healing process. Because titanium is osseointegrated, bone can grow right on top of it. This creates strong, long-lasting ties that are needed for implants to work in the long run.
People have been used in studies to show that titanium mixes better with living things than other materials. A study in the Journal of Biomedical Materials Research says that titanium surfaces help cells stick together and lower the number of inflammation reactions that happen. Because of this, they are perfect for use during important surgeries.
Mechanical Properties and Durability
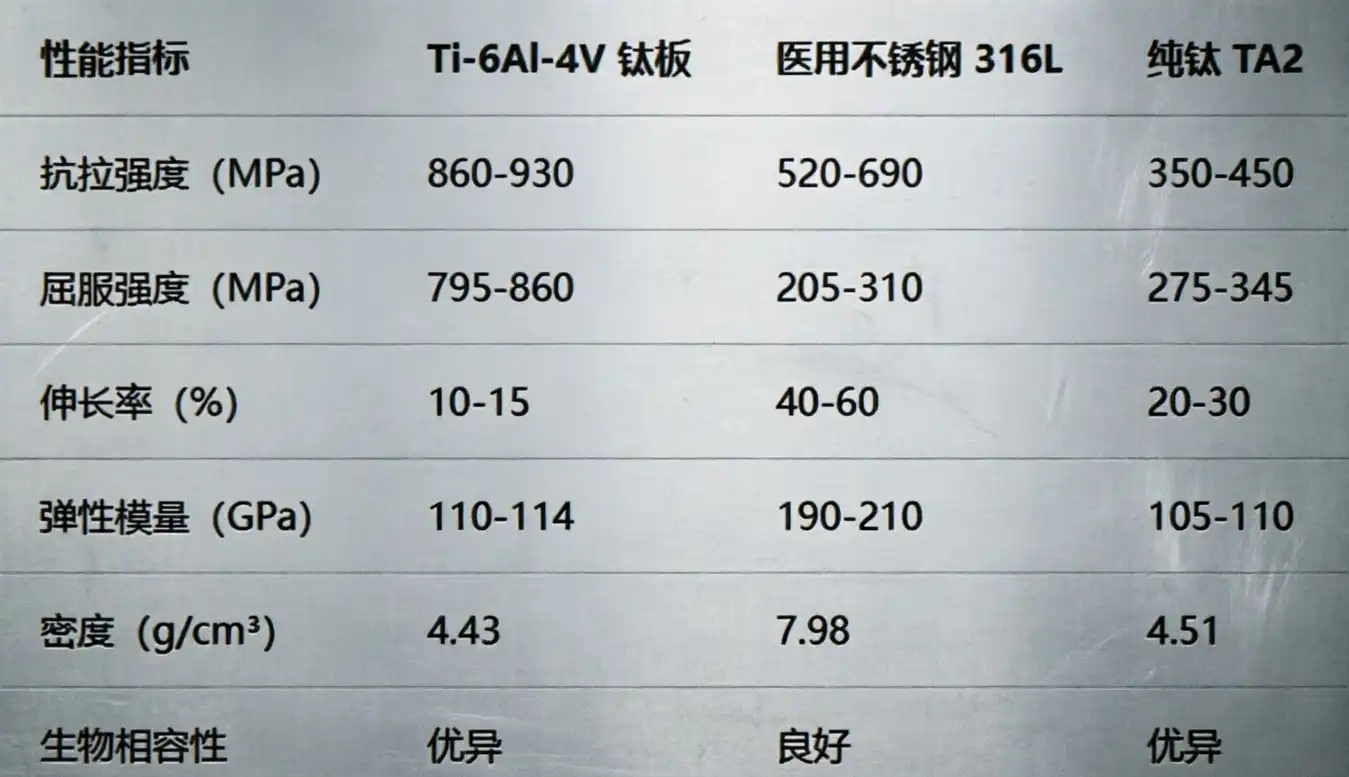
Titanium is light and strong, and it's better for medical use than most other metals. It supports the structure well without giving extra weight to the equipment that has been inserted. Pure types of titanium (Gr1, Gr2, Gr4) are bendy and simple to form. But titanium alloys, such as Ti-6Al-4V (Gr5), are harder and better for tough jobs.
Titanium can also help fight tiredness, which is another important medical benefit. In normal body conditions, stress corrosion cracking can happen in stainless steel. But titanium stays structurally strong through millions of heartbeats, so the implant lasts, and the patient is safe.
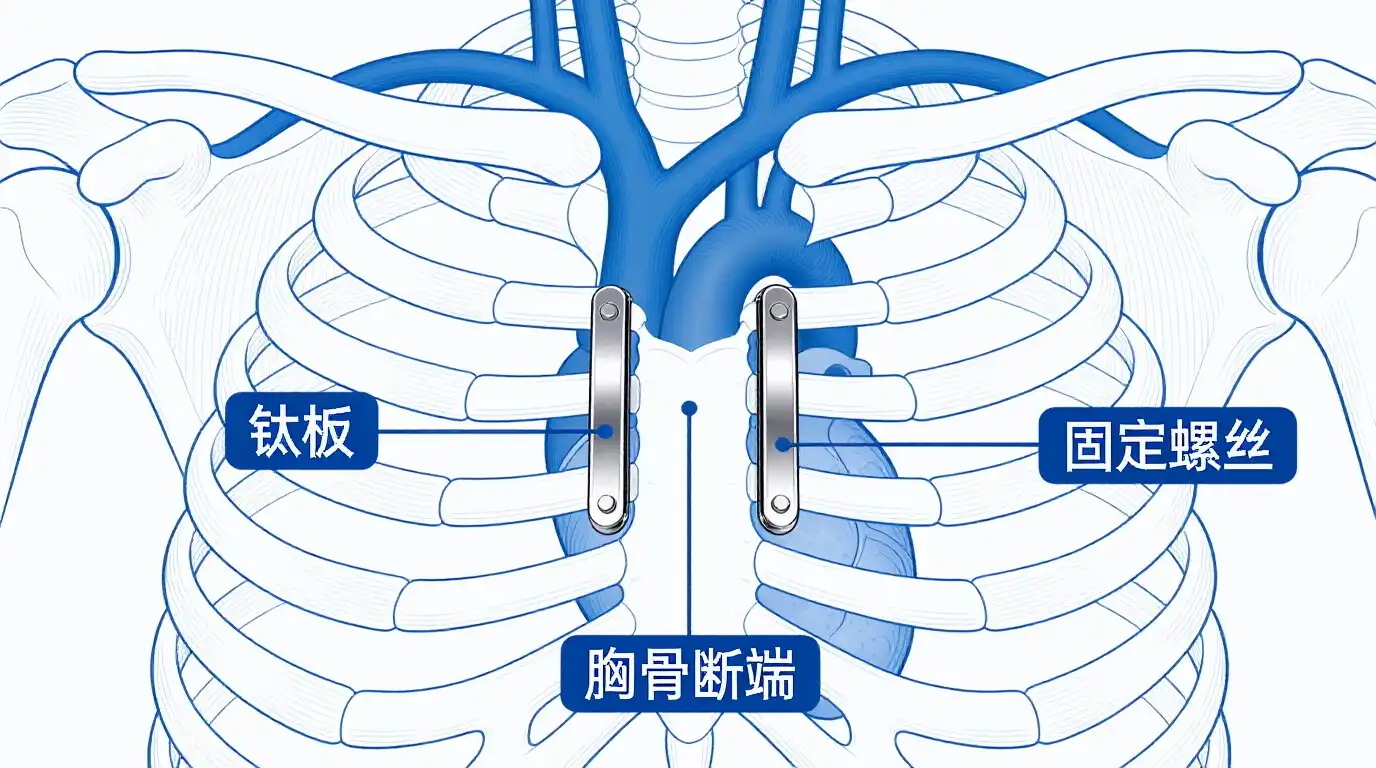
The Role of Titanium Plates in Open-Heart Bypass Surgery
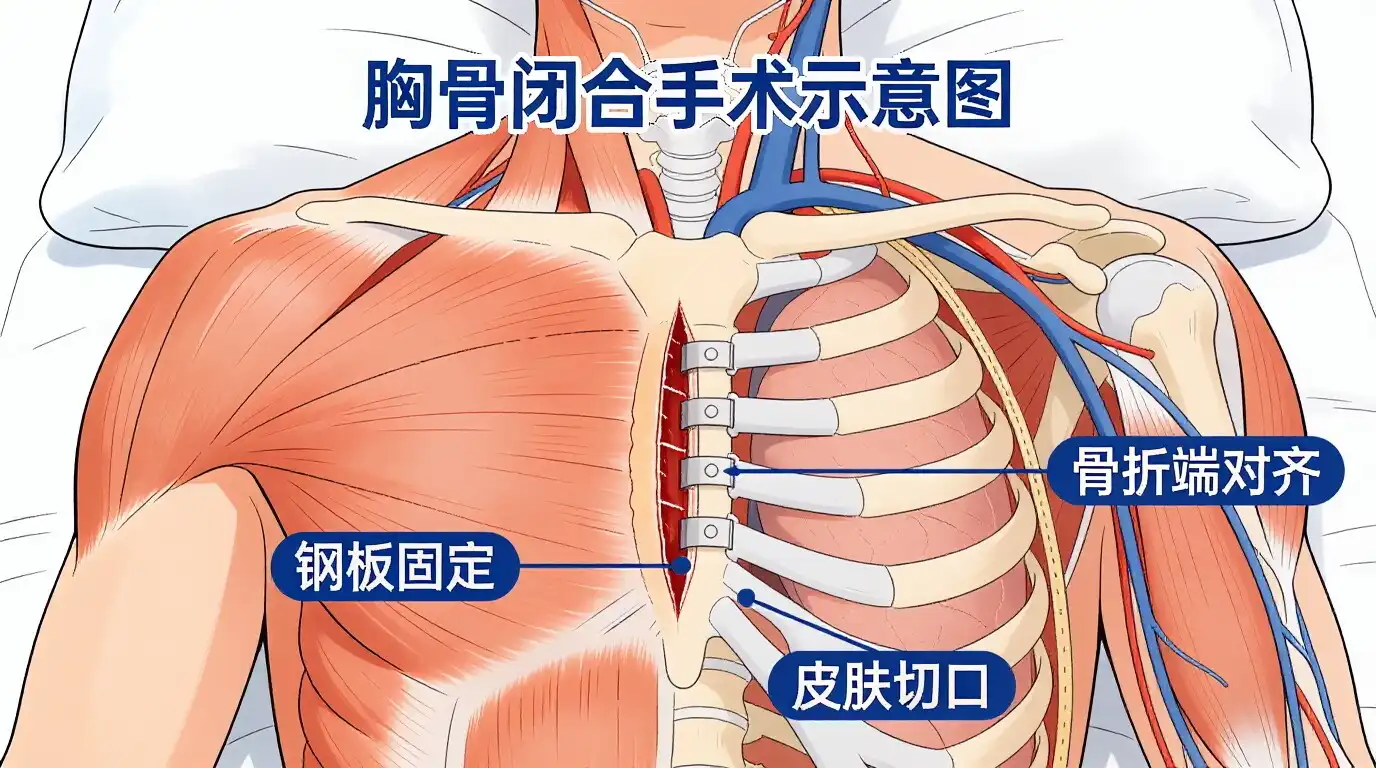
These days, heart surgeons use titanium plate devices to close the chest more and more after open-heart treatments. These plates are better at fixing things than regular wire cerclage, especially for people who are at high risk because their bones aren't very good or because they need difficult surgery.
Sternal Stabilization Systems
These days, titanium plate systems are used to close the sternum. These systems spread mechanical loads over bigger bone areas. This lowers the amount of stress that could break or fail to join the bone properly. What makes the SternaLock system so much better than regular wire fitting is the Ti-6Al-4V metal that is used to make it.
Three times as many problems happen after surgery with titanium sternal plating as with wire closing methods, according to research done at big heart centers. Patients can move around earlier and finish rehab programs faster because these devices keep them in place tightly.
Cardiac Device Integration
Most bypass grafts use biological or man-made channels, and the heart devices that go with them are often joined together by titanium. Because they don't rust and work well with electromagnetic fields, titanium metals are often used in the housings of pacemakers, defibrillators, and parts of cardiac assist devices.
Because it is pure titanium, it can be used with MRIs. This means that people with metal implants don't have to worry about their safety or getting bad pictures that could make the evaluation less accurate.
Procurement Considerations for Titanium Plates in Medical
Companies that make medical products and work for the military have unique problems when they need to get titanium parts for important projects. Grading standards, limits for size, and approval rules are important things to know before you buy something.
Grade Selection and Applications
Different kinds of titanium are used in different medical situations based on how well they work mechanically and with living things. These are the main types that are used to make medical tools:
- Grade 1 (Gr1): This is pure titanium that you can buy in shops. It is very flexible, which makes it ideal for medical tools and devices with thin walls that need to be shaped in complicated ways.
- Grade 2 (Gr2): This is the most popular type of medical titanium, grade 2 (Gr2). It's strong and bendy enough to be used in simple surgeries.
- Grade 4 (Gr4): This is pure titanium that is stronger and can be used for orthopedics and implants that can hold weight.
- Grade 5 (Ti-6Al-4V): This is the normal titanium metal. It is very strong and is used in the military and medical fields to make important buildings.
- Grade 23 (Ti-6Al-4V ELI): This is an extra-low interstitial form of Grade 5 that was created for medical implants that need to be more biocompatible.
Because of these grade differences, manufacturers can pick the best material for each job while still following the rules and keeping costs low.

Dimensional Specifications and Customization
To make sure that medical titanium plates fit and work right in surgery settings, the sizes of the plates need to be carefully controlled. The standard thickness ranges from 4 mm to 80 mm and can be used for different types of surgery, and the standard width ranges from 950 mm to 2500 mm, allowing special devices to be made.
Because the length can be increased up to 10,000 mm, big plates can be made for certain uses in heart and bone health. Companies that make medical equipment can get the most out of the materials they use while still meeting the needs of certain surgeries by offering custom sizes.

How to Manage Titanium Plate Specifications and Quality for Medical Use?
When it comes to medical titanium products, quality control rules are tighter than those used in other industries. These processes include thorough testing methods that look at both the biocompatibility and mechanical qualities. People who buy medical equipment can make sure they follow the rules if they know about these standards.
Processing and Surface Treatment Requirements
When medical titanium plates are made, they go through special steps that make them safe and improve their strength. In general, the steps are rolling, heating, leveling, cleaning, and finishing the surface. These steps get rid of flaws on the surface and stress groups.
The surface can be cut, polished, or acid-pickled, and all of these can be used for surgery. Because they don't rust as easily and germs won't stick to them, electropolished surfaces are great for long-term implantable devices that need to be biocompatible.

Certification and Traceability Standards
Providers of medical titanium must keep detailed records that make it possible to track materials all the way from where the raw materials are sourced to where the finished product is shipped. They need to show that they meet the requirements set by ASTM B265, ASTM F67, AMS 4911, and ASME SB265.
Mill test sheets are very important because they show the chemical makeup, the mechanical qualities, and the measurement check results. It is important for businesses that make medical equipment to follow the rules and make sure that the materials used in each batch are of the same quality.
Quality Assurance Testing Protocols
When the material is tested thoroughly, its tensile strength, yield strength, flexibility, and hardness are all checked to make sure they are good. Corrosion tests in fake body fluids have shown that it is stable over time in physiological circumstances.
Following ISO 10993 rules for biocompatibility testing ensures that titanium products are safe enough to be put into people. In order to make sure patients are safe, these studies look for inflammation, sensitivity, and damage to the whole body.

Future Trends and Innovations in Titanium Plates for Medical Applications
As new tools come out and people move around, the market for medical titanium is always changing to meet the needs of new surgery choices. People who work in shopping can guess what people will need in the future and find ways to stay ahead of the competition if they know about these trends.
Advanced Alloy Development
To use in medicine, scientists are creating new titanium alloy compositions that are better at being safe and having good mechanical properties. Beta titanium metals are more like bone in that they have lower elastic modulus values. This might mean that they don't protect against stress as well when used for orthopedics.
Additive manufacturing technologies let you make complicated titanium structures with inside shapes that aren't possible with older ways of making things. With these features, devices can be custom-made for each person, and gadgets can be made to work better.

Market Growth and Supply Chain Considerations
The medical titanium plate market is expected to be worth $2.4 billion by 2028. This is because more heart surgeries are being done, and people are living longer. People who work in buying and are in charge of titanium supply lines have both opportunities and problems because of this growth.
As the need for titanium grows, it becomes more and more important to have good ties with reliable sources. If a company has been making titanium goods for a long time and has quality standards, it will do well in the growing medical titanium market.
When procurement workers look for long-term partners, they should see what the seller can do, like keeping track of goods, providing special processing services, and having technical help resources on hand. Spreading source networks out across the world lowers the risks in the supply chain and makes sure that key materials are always available.
Conclusion
In open-heart bypass surgery, the titanium plates are very important. They are not used in bypass grafts themselves, but they are used in sternal closure systems and heart device parts. For important circulatory uses, medical-grade titanium metals are needed because they are biocompatible, don't rust, and are very strong. Buyers need to know about grade requirements, quality standards, and market trends in order to find the right titanium goods. As the market for medical titanium continues to grow, it's important to stay ahead of the competition in this niche area by making smart deals with providers and putting in place full quality management systems.
FAQ
Q1: What grades of titanium are suitable for cardiac surgery applications?
Most of the time, Grade 2 economically pure titanium and Grade 5 Ti-6Al-4V alloy are used in heart surgery. Grade 2 is very safe for living things and can be used for many medical tasks. Grade 5 is stronger and better for sternal closing systems and device housings that need to hold a lot of weight.
Q2: How do titanium plates compare to stainless steel for surgical applications?
Titanium plates offer superior biocompatibility, corrosion resistance, and strength-to-weight ratios compared to stainless steel alternatives. Additionally, titanium's paramagnetic properties ensure MRI compatibility, while stainless steel can create imaging artifacts and safety concerns during magnetic resonance procedures.
Q3: What certifications are required for medical titanium plates?
In order to be biocompatible and of good quality, medical titanium plates must meet the requirements set by ASTM F67, ASTM B265, and ISO 5832-2. Depending on the intended use and goal, it may be required to obtain FDA 510(k) clearance or CE marking.

Partner with Jucheng Titanium for Premium Medical Grade Materials
Medical device manufacturers seeking reliable titanium plate supplier partnerships can benefit from Jucheng Titanium's two decades of specialized experience and comprehensive quality management systems. Our medical-grade titanium plates meet stringent ASTM F67 and ASTM B265 standards while offering complete customization capabilities for thickness ranges from 4mm to 80mm and widths up to 2500mm.
Located in China's renowned Titanium Valley, our state-of-the-art 120,000 square meter facility maintains over 3,000 tons of titanium inventory, ensuring rapid delivery for urgent medical device production requirements. Our technical team collaborates with leading research institutions to develop innovative processing solutions that enhance biocompatibility and mechanical performance for critical surgical applications.
Contact our medical materials specialists at s4@juchengti.com to discuss your specific requirements and discover how our advanced processing capabilities can support your medical device manufacturing objectives.

References
1. Williams, D.F. "Titanium in Medicine: Material Science, Surface Science, Engineering, Biological Responses and Medical Applications." Springer-Verlag Berlin Heidelberg, 2001.
2. Niinomi, M. "Mechanical Biocompatibilities of Titanium Alloys for Biomedical Applications." Journal of the Mechanical Behavior of Biomedical Materials, Vol. 1, Issue 1, 2008, pp. 30-42.
3. Geetha, M., Singh, A.K., Asokamani, R., and Gogia, A.K. "Ti Based Biomaterials: The Ultimate Choice for Orthopedic Implants – A Review." Progress in Materials Science, Vol. 54, Issue 3, 2009, pp. 397-425.
4. Long, M. and Rack, H.J. "Titanium Alloys in Total Joint Replacement—A Materials Science Perspective." Biomaterials, Vol. 19, Issues 18-20, 1998, pp. 1621-1639.
5. Brunette, D.M., Tengvall, P., Textor, M., and Thomsen, P. "Titanium in Medicine: Material Science, Surface Science, Engineering, Biological Responses and Medical Applications." Engineering Materials Series, Springer, 2001.
6. Ryan, G., Pandit, A., and Apatsidis, D.P. "Fabrication Methods of Porous Metals for Use in Orthopedic Applications." Biomaterials, Vol. 27, Issue 13, 2006, pp. 2651-2670.









