What titanium alloy is used for surgical plates and pins?
Patients' implants must be composed of durable materials. Titanium alloy plates and pins, particularly Grade 5 (Ti-6Al-4V) and Grade 23 (Ti-6Al-4V ELI), are presently the best. These metals combine aluminium, vanadium, and titanium to form a substance strong enough to repair shattered bones yet light enough to avoid skin damage. Grade 23, or "extra low interstitial," has fewer contaminants. Its biocompatibility makes it the greatest choice for permanent implants in orthopaedic and orthodontic procedures, where the body must absorb foreign material without swelling or rejecting it.

Understanding Titanium Alloys for Surgical Plates and Pins
Medical-grade titanium is a set of precisely produced alloys intended to treat certain medical issues. Commercially pure titanium (Grades 1–4) resists corrosion; alloying modifies its medicinal properties.
The Core Medical-Grade Titanium Alloys
Grade 5 titanium alloy (Ti-6Al-4V) contains 6% aluminium and 4% vanadium. This material has a tensile strength of 895 MPa and a density of 4.43 g/cm³, about half that of stainless steel. This strength-to-weight advantage prevents stiff implants from breaking down bone and buffering stress. Grade 23 reduces oxygen, nitrogen, carbon, and iron considerably further. This makes it more flexible and wear-resistant, which is critical for devices loaded and unloaded millions of times during patient activity.
In a few cases, commercial Grade 2 pure titanium is chosen for rust resistance above strength. Unalloyed steel resists bodily fluids better, but it's too weak for load-bearing support plates. All output runs are the same since these materials are made to strict specifications, including ASTM B265, ASTM F67, and AMS 4911. Doctors need certain grain, thus hot-rolled plates are annealed, levelled, and then pickled in sequence.

Why Biocompatibility Matters in Surgical Implants
The defense system is always looking for threats, and putting metal into the body makes it look even more closely. Titanium's natural oxide layer forms in nanoseconds after being exposed to air. It acts as an inactive barrier to stop ions from entering nearby tissue. Long-term implantation studies that span decades have shown that titanium metals are not as harmful to cells as cobalt-chromium or even some types of stainless steel.
Studies in biomaterials journals show that Grade 23 titanium helps osseointegration, which is when bone directly bonds to implant surfaces, without the fibrous tissue entrapment that happens with lower-quality materials. This combination makes a strong base for trauma plates that hold broken bones together. Surgeons say that correction rates are lower when older materials are used instead of titanium. This means that fewer patients will get sick, and healthcare costs will go down.
Corrosion Resistance in the Body's Harsh Environment
The electrical environment, made up of chloride ions, proteins, and changing pH levels in blood and interstitial fluids, is very acidic. The inactive oxide film (mainly TiO₂) on titanium heals itself when it gets scratched, keeping its safety even when it's under a lot of mechanical stress. According to ASTM F2129 testing, titanium metals don't rust or pit in the same ways that stainless steel 316L does in the same situations.
When titanium alloy plates stay in place for years or even the rest of the patient's life, this stability is very important. Byproducts of metals breaking down can cause metallosis, which is tissue coloring and inflammation that needs to be fixed by removing the device. Titanium is very stable, so this risk is gone. This lets doctors focus on healing instead of worrying about the material failing.
Why Titanium Alloy Plates and Pins Are Preferred in Surgery?
Titanium anchoring systems are now used in operating rooms all over the world because they are better than older implant generations at fixing problems. The benefits go beyond just figuring out the strength; they also affect how well the surgery goes and how well the patient does.
Lightweight Strength Reduces Patient Burden
A Grade 5 titanium wrist fracture fixation plate weighs about 40% less than a similar stainless steel device, but it provides the same amount of muscular support or more. This weight loss makes the strange body feeling that patients say they feel after surgery less severe. When athletes and workers go back to competition or hard work, the affected leg is less tired, which is directly related to the lower implant mass.

The modulus of elasticity for Ti-6Al-4V is about 110 GPa, which is lower than the modulus of elasticity for stainless steel, which is 200 GPa. This mechanical resemblance lets the healing fracture spot naturally spread the load. When implants are too stiff, they protect bone from bodily stress, which causes resorption and makes the fix less strong. Titanium's "Goldilocks" stiffness helps bones grow and change healthily while keeping them in place during the most important time for healing.
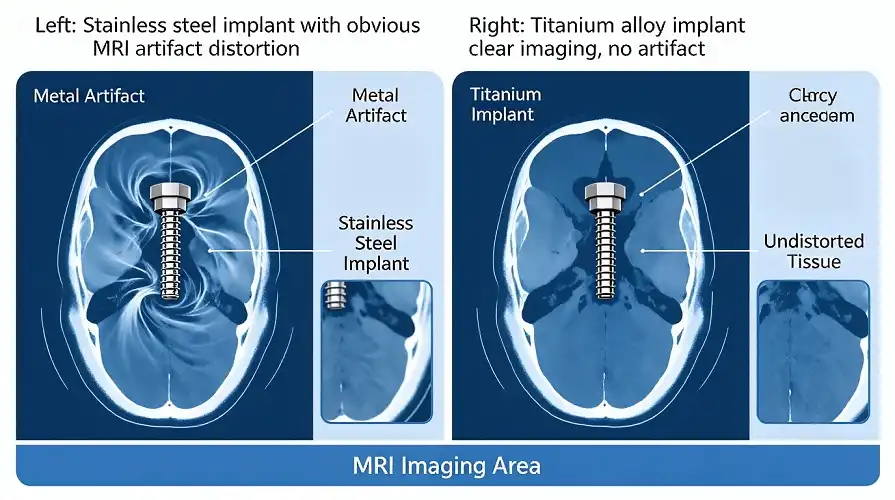
MRI Compatibility Opens Diagnostic Possibilities
For checking out soft tissue injuries, tumors, and arterial diseases, magnetic resonance imaging is now a must. Some types of ferromagnetic metals, like some grades of stainless steel, make huge image artifacts, which are dark holes that hide tissue near implants. Even worse, the magnetic field can twist or heat ferromagnetic items, which can be dangerous for patients.
Titanium's paramagnetic features make artifacts very small, which lets doctors see the tissues around implants. With a titanium mandibular replacement plate, a brain MRI can be done without taking off any hardware. With older steel implants, the patient would need to have the implant removed or have to use less accurate imaging methods. As medical imaging gets better, this connectivity can't be changed.
Material Comparison Reveals Clear Advantages
When you compare titanium to other materials, you can see why purchasing managers at companies that make medical devices choose it over others, even though it costs more:
- Stainless Steel 316L: Strong and cheap; however, nickel content causes allergic responses in sensitive persons. In long-term implantation, corrosion resistance is poor, and MRI incompatibility hinders diagnosis.
- Cobalt-Chromium Alloys: Excellent wear resistance for joint replacements, but stiffness causes fracture fixation stress shielding. Manufacturing complexity and material cost exceed titanium.
- Emerging biodegradable magnesium alloys dissolve in the body, avoiding removal procedures. Clinical unpredictability is caused by premature deterioration before fracture repair and uneven mechanical characteristics during absorption.
Orthopedic trauma surgeons treating complicated pelvic fractures increasingly specify titanium reconstruction plates because they can handle the high loads that these body parts experience when they bear weight. Craniofacial surgeons reconstructing orbital floors after trauma rely on thin titanium mesh that fits the shape of the bone while keeping the structure strong. Real-world applications validate titanium's clinical superiority backed by decades of surgical outcomes data.
Manufacturing and Machining of Titanium Alloy Surgical Plates and Pins
Making medical-grade titanium parts requires a level of accuracy that goes far beyond what is needed in general industry settings. Each step in the process changes the end implant's surface features, mechanical qualities, and biological performance.
Advanced Machining Techniques for Surgical Precision
Titanium is very hard to machine because it work-hardens quickly, doesn't conduct heat well, and clashes with tool materials when heated up. To keep tolerances tight, companies that make medical devices use special carbide or polycrystalline diamond cutting tools with shapes that are designed. Fixation plates made by CNC milling centers have holes that are placed within ±0.05 mm of accuracy, which is very important for aligning screws during surgery.
Surface finishing determines how bone and soft tissue react to inserted titanium alloy plates. Acid pickling removes surface dirt and builds up a smooth metal layer. More polishing may be done on machined surfaces to reduce bacterial adhesion points, particularly important for implants penetrating skin, like external fixation pins. Some manufacturers apply grit blasting to create controlled surface roughness, promoting osseointegration on bone-contact surfaces while keeping soft-tissue-facing areas smooth.
Heat Treatment for Optimal Mechanical Properties
Solution treatment and aging cycles modify the alpha-beta microstructure in Grade 5 and Grade 23 alloys, tailoring strength and ductility. Plates destined for high-stress applications undergo heat treatment, producing ultimate tensile strength approaching 930 MPa. The annealing process reduces residual stresses from forming operations, preventing unexpected deformation during sterilization or surgical contouring.
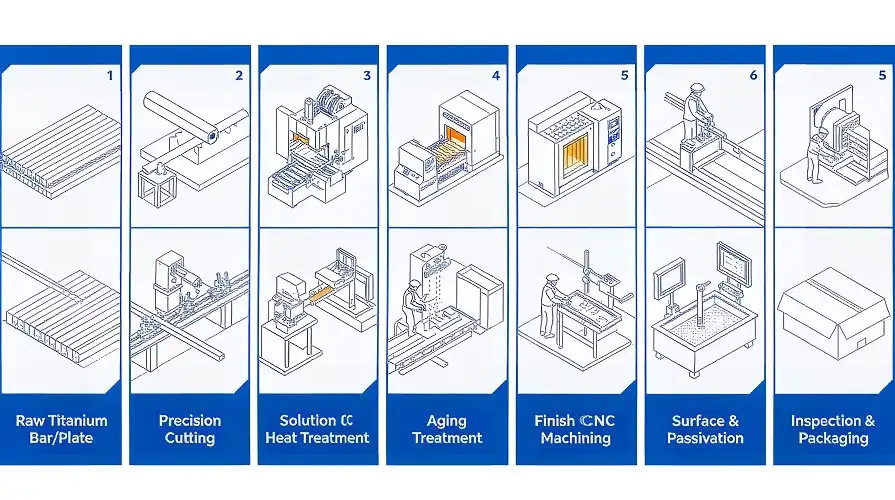
Quality control during heat treatment involves precise temperature and atmosphere control. Exposure to excess oxygen or nitrogen at elevated temperatures increases interstitial content, degrading the Grade 23 "extra low interstitial" specification. Vacuum or inert gas furnaces protect material purity. Metallurgical testing, including microscopy and tensile testing on production samples, verifies heat treatment effectiveness before batches receive certification.
Certifications and Traceability Standards
Medical device laws globally need complete raw material-to-finished product documentation. ASTM F136 titanium suppliers issue mill certifications for each ingot documenting chemical composition, mechanical test findings, and heat treatment parameters. This documentation chain includes production, inspection, and sterilisation reports from medical device makers.
A manufacturer's ISO 13485-certified quality management system fulfils medical device standards. US FDA approval needs 510(k) clearance or Premarket Approval for safety and efficacy. MDR compliance requires Notified Body evaluation and CE certification. These regulations safeguard patients but complicate titanium plate procurement for device makers.
Plates made to these requirements include laser-etched lot numbers for full traceability. Manufacturers can target manufacturing runs for post-market monitoring recalls rather than market withdrawals. Traceability supports patient safety and manufacturer responsibility.
Procurement Considerations for Titanium Alloy Plates and Pins in Medical Applications
Getting titanium for medical uses is very different from getting general metals for industry. Device manufacturers must balance regulatory compliance, supply chain reliability, and cost management while never compromising quality.
Evaluating Global Suppliers and Certifications
The global titanium supply chain includes major producers like VSMPO-AVISMA in Russia, ATI Specialty Materials in the United States, and multiple suppliers in China's Baoji region—known as "Titanium Valley" for its concentration of expertise. Evaluating suppliers requires examining multiple dimensions beyond price quotations.
Certification portfolios matter enormously. Suppliers should hold ISO 13485 registration demonstrating medical device quality systems. Material certifications must confirm compliance with ASTM B265 for titanium plate specifications and ASTM F136 or F67 for surgical implant materials. Request certificates from independent testing laboratories verifying chemical composition and mechanical properties rather than relying solely on supplier self-certification.
Supplier longevity and industry reputation provide confidence in sustained quality for titanium alloy plates. Manufacturers operating for decades have refined processes and trained personnel essential for consistent production. Newer entrants may offer attractive pricing but lack the institutional knowledge to prevent quality excursions. Reference checks with existing customers reveal supplier responsiveness when issues arise—a critical consideration given the high stakes of medical device manufacturing.
Customization and Order Specifications
Standard titanium plates arrive in dimensions ranging from 4mm to 80mm in thickness, widths between 950mm and 2500mm, and lengths up to 10,000mm. Medical device applications often require custom sizing to optimize material utilization and minimize machining waste. Suppliers offering cutting, annealing, and surface finishing services add value by delivering material closer to the final geometry.
Specifying the correct grade proves critical. Grade 5 (Ti-6Al-4V) serves most orthopedic fixation devices requiring high strength. Grade 23 becomes essential for permanent implants where superior ductility and fatigue resistance justify modestly higher costs. Grade 2 commercially pure titanium suits applications prioritizing corrosion resistance over maximum strength, such as certain spinal fusion cages.
Order quantities influence pricing and lead times significantly. Minimum order quantities for medical-grade material may exceed general industrial purchases due to specialized quality control requirements. Lead times extending 8-16 weeks aren't uncommon as suppliers coordinate production campaigns, conduct testing, and prepare certification documentation. Strategic buyers maintain safety stock or establish framework agreements guaranteeing material availability for production surges.
Strategic Supplier Partnerships for Supply Chain Resilience
Medical device manufacturers increasingly recognize that single-source supply chains create vulnerability. Geopolitical disruptions, natural disasters, or supplier financial instability can halt production with devastating business impacts. Dual sourcing from geographically diverse suppliers provides insurance, though managing multiple material qualifications increases regulatory burden.
Long-term partnership agreements benefit both parties. Suppliers gain volume visibility, enabling capacity planning investments. Buyers secure preferential pricing, priority allocation during shortages, and collaborative problem-solving when technical challenges emerge. Some agreements include vendor-managed inventory, where suppliers maintain consignment stock at the buyer's facility, reducing working capital requirements while ensuring material availability for titanium alloy plates.
Bulk purchasing leverages volume for cost reduction but requires careful inventory management. Titanium doesn't degrade in storage, yet tying capital in excess inventory creates opportunity costs. Negotiating flexible delivery schedules—purchasing annual volumes with quarterly shipments—balances cost savings against inventory holding expenses.

Conclusion
Titanium alloys, particularly Grade 5 and Grade 23, dominate surgical plate and pin applications because they solve the most demanding challenges in medical implantation. The combination of biocompatibility, corrosion resistance, appropriate mechanical properties, and MRI compatibility creates unmatched clinical performance. Manufacturing these materials to medical device standards requires sophisticated processing, rigorous quality control, and comprehensive certification—factors that make supplier selection critical for device manufacturers. As surgical techniques advance and patient expectations rise, titanium's role will expand further. Procurement professionals who understand the technical nuances, regulatory landscape, and supply chain dynamics position their organizations to deliver life-changing medical devices built on this remarkable material's foundation.
FAQ
Q1: What grade of titanium alloy is best for surgical plates?
Grade 5 (Ti-6Al-4V) and Grade 23 (Ti-6Al-4V ELI) represent the optimal choices for surgical plates and fixation devices. Grade 5 provides excellent strength for trauma applications, while Grade 23's extra low interstitial content offers superior ductility and fatigue resistance for permanent implants. The selection depends on specific clinical requirements—high-load applications favor Grade 5, whereas implants subjected to cyclic loading or remaining permanently benefit from Grade 23's enhanced properties.
Q2: How does titanium compare to stainless steel for medical implants?
Titanium surpasses stainless steel in biocompatibility, corrosion resistance, and MRI compatibility. The lower density reduces implant weight by approximately 40%, decreasing patient discomfort. Titanium's modulus of elasticity better matches bone, reducing stress shielding that weakens healing fractures. Stainless steel costs less initially, but higher revision rates from corrosion and nickel sensitivity often negate savings. Titanium's paramagnetic properties allow post-operative MRI imaging without artifact or safety concerns that plague ferromagnetic stainless steel grades.
Q3: What certifications should I look for when sourcing medical titanium?
Verify suppliers hold ISO 13485 certification for medical device quality management systems. Material must conform to ASTM F136 (for Grade 5 and 23 surgical implants) or ASTM F67 (for unalloyed titanium). ASTM B265 covers general titanium plate specifications. Request mill certificates documenting chemical composition and mechanical testing. Depending on target markets, confirm FDA registration for United States distribution or CE marking under the European Medical Device Regulation. Traceability documentation linking finished products to certified raw material lots is mandatory for regulatory compliance.
Partner with Jucheng Titanium for Premium Medical-Grade Titanium Alloy Plates

Selecting a reliable titanium alloy plate supplier directly impacts your product quality and market success. Jucheng Titanium brings over 20 years of deep expertise in titanium material processing, maintaining 3,000 tons of inventory for rapid delivery that matches your production schedules. Our medical-grade titanium plates conform to ASTM B265, ASTM F67, and AMS 4911 standards, available in Grades 1, 2, 4, 5, 7, 9, and 12 with customizable dimensions (thickness 4-80mm, width 950-2500mm, length up to 10,000mm). Hot-rolled, annealed, and finished through precision machining, acid pickling, or polishing, our materials meet the stringent requirements medical device manufacturers demand. Collaboration with Northwest Nonferrous Metals Research Institute and recognition as a national-level specialized "little giant" enterprise underscore our technical capabilities. Contact our team at s4@juchengti.com to discuss your specific requirements—our engineering support and flexible customization ensure you receive exactly what your surgical implant applications require.
References
1. Long, M., & Rack, H.J. (1998). Titanium alloys in total joint replacement—a materials science perspective. Biomaterials, 19(18), 1621-1639.
2. Niinomi, M. (2008). Mechanical biocompatibilities of titanium alloys for biomedical applications. Journal of the Mechanical Behavior of Biomedical Materials, 1(1), 30-42.
3. Geetha, M., Singh, A.K., Asokamani, R., & Gogia, A.K. (2009). Ti based biomaterials, the ultimate choice for orthopaedic implants—A review. Progress in Materials Science, 54(3), 397-425.
4. Rack, H.J., & Qazi, J.I. (2006). Titanium alloys for biomedical applications. Materials Science and Engineering: C, 26(8), 1269-1277.
5. Banerjee, D., & Williams, J.C. (2013). Perspectives on titanium science and technology. Acta Materialia, 61(3), 844-879.
6. Liu, X., Chu, P.K., & Ding, C. (2004). Surface modification of titanium, titanium alloys, and related materials for biomedical applications. Materials Science and Engineering: R: Reports, 47(3-4), 49-121.









